Speaker
Description
Binary liquid mixtures of alcohols and ethers are of importance as potential biofuels or additives for internal combustion engines,[1] and have also attracted fundamental interest as model liquid systems containing one component (the alcohol) that can strongly self-associate through hydrogen bonding (HB), and one that cannot self-associate via HBs (ether), yet can interact strongly with the former as HB acceptor.[2] The excess thermodynamic properties of these mixtures, specifically the excess molar enthalpies and volumes (HE and VE), have been extensively measured.[1-3] Butanol isomer + di-n-butyl ether (DBE) binary mixtures, in particular, show interesting volumetric differences, with VE changing from negative (1- and iso-butanol) to positive (2- and tert-butanol) with increasing butanol alkyl group branching. Representative 1- and 2-butanol + DBE mixtures were studied, for the first time, by atomic-resolution classical Molecular Dynamics (MD) computer simulations. The simulations reveal decided differences in the degree of self-association of the two butanol isomers and support existing interpretations of the HE and VE in a general sense, but also suggest that more subtle differences in H-bonded topologies may contribute significantly to the anomalous volumetric properties of these mixtures.[4]
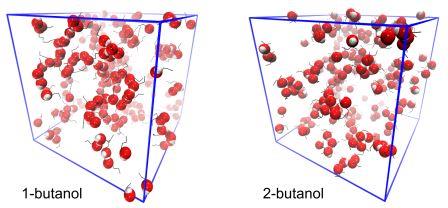
Fig. 1. MD computer simulation configurations of equimolar mixtures of 1- or 2-butanol with DBE. Ether molecules are omitted and hydroxyl (-OH) groups shown using a space-filling (red and white spheres) representation in order to highlight the differences in HB topologies. Simulation cell edges are shown in blue.
References:
[1] (a) Rezanova, E. N.; Kammerer, K.; Lichtenthaler, R. N. J. Chem. Eng. Data 1999, 44, 1235-1239. (b) Alaoui, F. E. M.; Montero, E. A.; Brazile, J. -P.; Aguilar, F.; Boned, C. J. Chem. Thermodynamics, 2011, 43, 1768-1774.
[2] (a) Motoyama, I.; Jarboe, C. H. J. Phys. Chem. 1967, 71 (8), 2723-2726. (b) Pathak, G.; Pradhan, S. Proc. Indian Acad. Sci. (Chem. Sci.) 1988, 100 (6), 519-523. (c) Lechter, T. M.; Govender, P. U. Fluid Phase Equilibria, 1997, 140, 207-220.
[3] (a) Patil,K. R.; Pathak, G.; Pradhan, S. D. Proc. Indian Acad. Sci. (Chem. Sci.) 1989, 101 (5), 443-447. (b) Kammerer, K.; Licthenthaler, R. N. Thermochim. Acta 1998, 310 (1-2), 61-67. (c) Bernazzani, L.; Carosi, M. R.; Duce, C.; Gianni, P.; Mollica, V. J. Solution Chem. 2006, 35 (11), 1567-1585.
[4] Engelbrecht, L.; Farris, R.; Vasiliu, T.; Demurtas, M.; Laaksonen, A.; Piras, A.; Cesare Marincola, F.; Porcedda, S.; Mocci. F. Submitted for publication.
